Alzheimer’s disease, the leading cause of dementia, has long puzzled researchers with its complex origins. A new study proposes a “unifying theory” suggesting the disease develops when amyloid-beta and tau proteins compete for space inside brain cells, rather than acting as independent causes. This finding could reshape how we understand and treat the devastating condition, which currently affects millions worldwide.
The Long-Standing Mystery of Amyloid and Tau
For decades, scientists have debated the role of two primary hallmarks of Alzheimer’s: amyloid-beta plaques (sticky clumps outside neurons) and tau tangles (twisted fibers inside neurons). Both appear in the brains of Alzheimer’s patients, but whether they cause the disease or are merely symptoms has remained unclear. Some research suggests amyloid-beta builds up years before symptoms appear, while others emphasize tau tangles as a stronger predictor of cognitive decline.
The debate is crucial because treatment strategies have largely focused on clearing one or the other, with limited success. If both are effects of something else, those therapies might be misdirected.
The Competition Theory: Amyloid-Beta Disrupts Tau Function
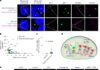
Researchers at the University of California, Riverside, found evidence that amyloid-beta actively displaces tau from its crucial role in stabilizing brain cells. Tau proteins normally support the internal structure of neurons (microtubules), but when detached, they form tangles, disrupting cell function.
In laboratory experiments, the team observed that amyloid-beta peptides preferentially bind to microtubules, effectively stealing the spots normally occupied by tau. This competition suggests that amyloid-beta doesn’t simply accumulate; it interferes with essential cellular processes.
“Our work shows amyloid beta and tau compete for the same binding sites on microtubules, and that [amyloid-beta] can prevent tau from functioning correctly,” says study author Ryan Julian.
Why This Matters: A Shift in Therapeutic Focus
This discovery has significant implications. If amyloid-beta drives tau dysfunction, then the primary toxicity might stem from destabilized microtubules, not simply the presence of plaques or tangles. This challenges existing therapies focused on clearing amyloid-beta, which have yielded disappointing results in clinical trials.
The research suggests that future treatments should prioritize protecting microtubule stability rather than solely targeting protein buildup. Interestingly, preliminary animal studies hint at lithium’s potential protective effect by stabilizing microtubules, offering a possible avenue for exploration.
The Road Ahead: From Lab to Clinic
While promising, these findings are based on purified protein studies, and translating them to the complex environment of living brains is a major challenge. The next step involves confirming these interactions within neurons and exploring how to prevent amyloid-beta from disrupting tau function in vivo.
However, this study clarifies many previously conflicting results, providing a more coherent model for Alzheimer’s development. If validated, it could redirect research toward therapies that preserve the integrity of brain cells, potentially offering a new path toward treating a disease with no known cure.