For decades, scientists have known that cytotoxic T lymphocytes are the immune system’s elite strike force. These cells hunt down and destroy cancerous or infected cells with remarkable precision. However, the exact mechanics of how they deliver their lethal blow—without harming surrounding healthy tissue—remained largely invisible to researchers.
A new study by scientists at the University of Geneva (UNIGE) and Lausanne University Hospital (CHUV) has changed that. By using a novel imaging technique called cryo-expansion microscopy, they have captured the first high-resolution, three-dimensional view of these killer cells in action inside human tumors. Their findings, published in Cell Reports, reveal the intricate molecular architecture that allows T cells to function, offering new clues for improving cancer immunotherapy.
The Immune Synapse: A Precision Strike
The key to a T cell’s effectiveness lies in a structure called the immune synapse. When a T cell identifies a target, it attaches to the cancer cell and forms this specialized contact point. Through the synapse, the T cell releases toxic molecules that trigger the death of the target cell.
This process is incredibly delicate. It must be powerful enough to destroy the threat but controlled enough to leave neighboring healthy cells intact. While the general mechanism is well understood, observing the nanometer-scale details of this interaction inside intact human cells has been nearly impossible. Traditional imaging methods often require preserving samples in ways that distort or destroy these fragile structures, forcing scientists to choose between high resolution and biological accuracy.
Freezing the Action: Cryo-Expansion Microscopy
To overcome these limitations, the research team, supported by the ISREC Foundation TANDEM program, turned to cryo-expansion microscopy (cryo-ExM). This technique offers a unique solution to the resolution-versus-preservation dilemma.
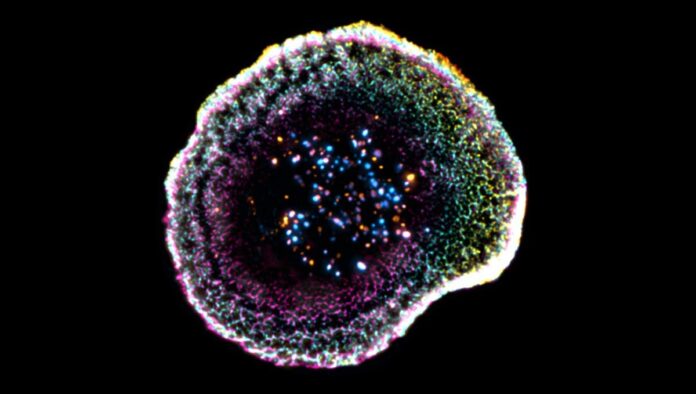
“This technique involves instantaneously freezing cells at very high speed, placing them in a so-called vitreous state, where water solidifies without forming crystals and thus faithfully preserves biological structures. The samples are then physically expanded using an absorbent hydrogel, making it possible to observe their internal organization with great precision while maintaining their near-native architecture,” explains Virginie Hamel, Senior Lecturer at UNIGE.
By freezing the cells instantly, the researchers avoided the ice crystals that typically damage cellular structures. They then expanded the sample physically, effectively zooming in on the cellular machinery without losing its natural shape.
Hidden Structures Revealed
This new lens revealed previously unseen details about how T cells operate:
- The Membrane Dome: At the point of contact, the T cell’s membrane forms a distinct dome-like structure. Florent Lemaître, the study’s first author, notes that this shape appears linked to adhesion interactions and the cell’s internal organization, suggesting a mechanical component to how T cells “lock on” to their targets.
- Variable Granules: The team examined cytotoxic granules—the containers holding the toxic molecules—in unprecedented detail. They found that these granules are not uniform; they vary in structure, sometimes containing single or multiple “cores” where active molecules are concentrated. This variability may influence how efficiently a T cell can kill its target.
Seeing the Battlefield Inside Tumors
Perhaps the most significant advancement is that this technique works not just on isolated cells, but on actual human tumor tissues.
“We extended this approach to human tumor tissues, making it possible to directly observe T lymphocytes infiltrating tumors and their cytotoxic machinery at the nanometer scale,” says Benita Wolf, Chief Resident and Associate Researcher at CHUV. “This allows us to study immune responses directly in their clinical context and to better understand the mechanisms that determine their effectiveness.”
Why This Matters for Cancer Treatment
Current immunotherapies aim to boost the body’s natural ability to fight cancer, but they do not work for everyone. Understanding the physical and molecular details of how T cells interact with tumors is crucial for improving these treatments.
By providing a clear, three-dimensional map of the immune synapse in its natural environment, this research helps answer critical questions:
* What structural features make a T cell more effective?
* Why do some immune responses fail to penetrate tumors?
* How can therapies be designed to support or enhance these natural killing mechanisms?
In summary, this breakthrough moves beyond theoretical models to provide a direct visual understanding of the immune system’s most powerful weapon. By revealing the precise architecture of T cell attacks, scientists are one step closer to designing more effective, targeted cancer therapies that harness the body’s own defenses.